 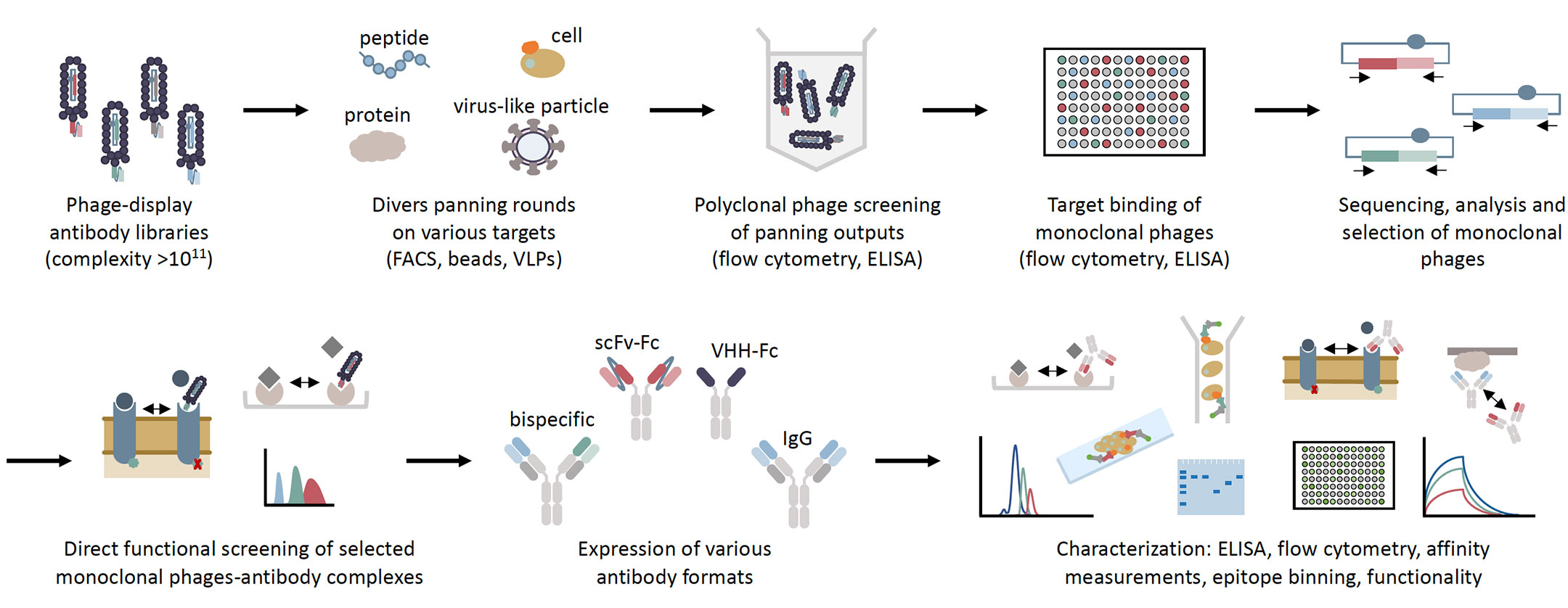
DENV NS5 protein inhibits IFN-I signaling by mediating proteasome-dependent STAT2 degradation. Antagonism of type I interferon (IFN-I) signaling is a crucial mechanism of DENV immune evasion. An improved understanding of DENV immune evasion is pivotal for the rational development of anti-DENV therapeutics. Despite the prevalence and potential severity of DF and DHF, there are no approved vaccines or antiviral therapeutics available. 
We discuss potential implications of these findings for mindset-based interventions aimed at reducing test anxiety.Īn estimated 50 million dengue virus (DENV) infections occur annually and more than forty percent of the human population is currently at risk of developing dengue fever (DF) or dengue hemorrhagic fever (DHF). Students’ intelligence mindset at pretest was a significant predictor of test anxiety at posttest, and this relationship was mediated by self-reported effort regulation. Effort regulation was measured as self-reported judgements of persistence in the face of difficulty and test anxiety was measured on a problem by problem basis during a laboratory physics assessment. We tested our model as an exploratory post-hoc analysis in a small sample of introductory physics students reporting psychological threat in a laboratory study. We propose that students’ beliefs about effort regulation may partially mediate the relationship between intelligence mindsets and test anxiety. Prior research has proposed interventions aimed at changing students’ implicit beliefs about intelligence (intelligence mindset) to help reduce students’ test anxiety, but results have been mixed and the mechanisms underlying this relationship are unclear. Test anxiety affects a sizable proportion of college students, especially in competitive STEM fields. These observations provide insight into the pathogenesis of antibody-enhanced dengue disease and identify a novel strategy for the design of therapeutic antibodies against dengue. In contrast, a genetically engineered antibody variant (E60-N297Q) that cannot bind FcγR exhibited prophylactic and therapeutic efficacy against ADE-induced lethal challenge. Passive transfer of a high dose of serotype-specific antibodies eliminated viremia, but lower doses of these antibodies or cross-reactive polyclonal or monoclonal antibodies all enhanced disease in vivo even when antibody levels were neutralizing in vitro. Antibody-enhanced lethal disease featured many of the hallmarks of severe dengue disease in humans, including thrombocytopenia, vascular leakage, elevated serum cytokine levels, and increased systemic viral burden in serum and tissue phagocytes. Analogous to human infants who receive anti-DV antibodies by transplacental transfer and develop severe dengue disease during primary infection, we show here that passive administration of anti-DV antibodies is sufficient to enhance DV infection and disease in mice using both mouse-adapted and clinical DV isolates. However, despite decades of investigation, no in vivo model for antibody enhancement of dengue disease severity has been described. Serotype cross-reactive antibodies facilitate DV infection of myeloid cells in vitro by promoting virus entry via Fcγ receptors (FcγR), a process known as antibody-dependent enhancement (ADE). 
Immunity to one of the four dengue virus (DV) serotypes can increase disease severity in humans upon subsequent infection with another DV serotype. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
